- Accueil
- ylm tablette
- PDF) Development and validation of zero and first-order derivative area under curve spectrophotometric methods for the determination of aripiprazole in bulk material and tablets
PDF) Development and validation of zero and first-order derivative area under curve spectrophotometric methods for the determination of aripiprazole in bulk material and tablets
4.7 (768) · € 16.00 · En Stock
The proposed experiments explain simple, precise, specific and accurate UV spectrophotometry methods for the estimation of Aripiprazole in bulk and pharmaceutical formulation. Aripiprazole is a recent second generation atypical anti-psychotic drug

Factorial approach for the optimization and development of stability indicating study of the contraceptive suspension for injection(Depo-Provera®) - ScienceDirect
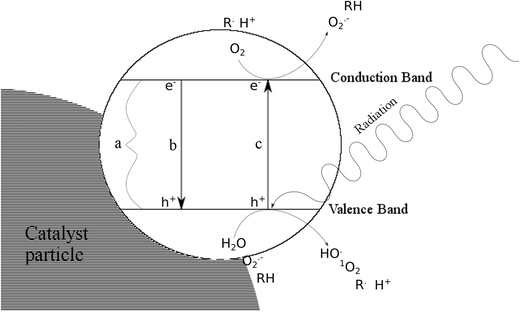
Studies on photodegradation process of psychotropic drugs: a review

Chaotropic chromatography method development for the determination of aripiprazole and its impurities following analytical quality by design principles - Rmandić - 2020 - Journal of Separation Science - Wiley Online Library

PDF] Validated spectrophotometric quantification of aripiprazole in pharmaceutical formulations by using multivariate technique.
Sustainable eco-friendly ratio-based spectrophotometric and HPTLC-densitometric methods for simultaneous analysis of co-formulated anti-migraine drugs with overlapped spectra, BMC Chemistry

PDF) Development and Validation of Uv-Visible Spectrophotometric Method for Estimation of Tadalafil in Bulk and Formulation
Spectrofluorometric quantitative analysis of aripiprazole based on quenching of natural derived carbon quantum dots in spiked human plasma

Quantitative estimation of molnupiravir by UV- Spectrophotometric method - IJPCA

Development and Validation of UV‐ Spectrophotometric Method for Determination of Naftopidil in Bulk by Shirley Wang - Issuu

Estimation of Levocetirizine in Bulk and Formulation by First Order Derivative Area Under Curve UV-Spectrophotometric Methods, PDF, Detection Limit

PDF) Development and Validation of UV Spectrophotometric Estimation of Ibuprofen in Bulk and Tablet Dosage Form Using Area under Curve Method
Validated Area Under Curve and Zero Order Spectroscopic Methods for Estimation of Agomelatine in Bulk and Pharmaceutical Tablet Dosage Form – Biomedical and Pharmacology Journal












